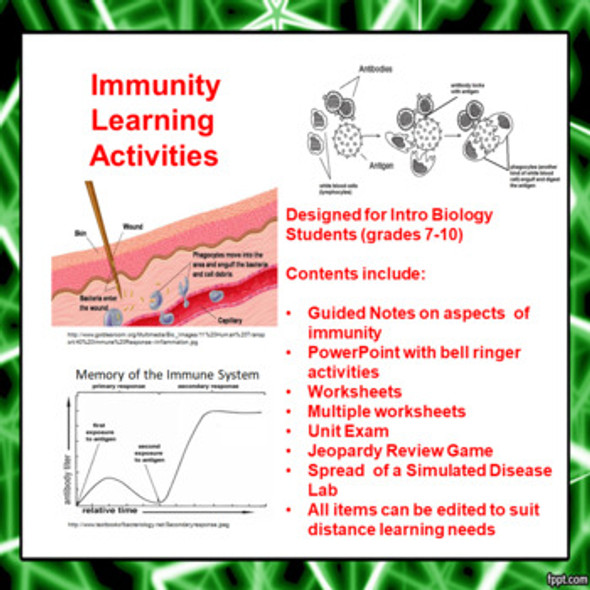
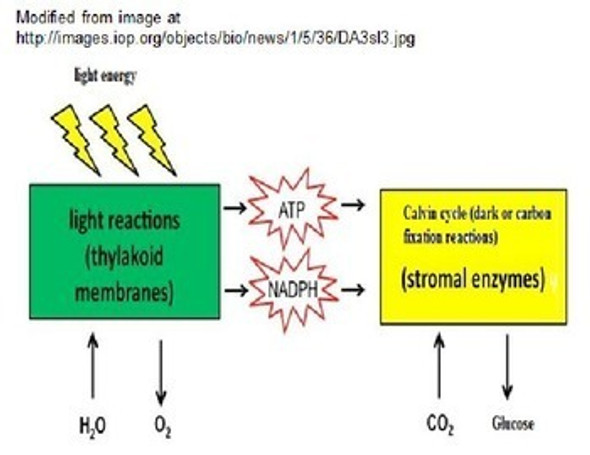
Physical Behavior of Matter Learning Activities (Distance Learning)
- Bulk Pricing:
- Buy in bulk and save
- Contributor:
- Monday's Rescue
- Grade Level:
- 9-12
- Product Type:
- Learning Package Bundle (Notes, PowerPoint, Lab, Homework)
- File Type:
- doc, pdf, ppt
- Pages:
- 43
- Answer Key:
- Included with all but the lab
Description
This zip file contains many different activities (43 pages of student handouts and 1 PowerPoint with a total of 59 slides) which can be used to compose a unit for chemistry students involving the Physical Behavior of Matter. Topics in this unit include temperature conversions, phases of matter, changes of state, specific heat, heat of fusion, heat of vaporization and computations involving these topics. Additional topics include the Gas Laws and computations involving these (Boyle's Law, Charles' Law, the Combined Gas Law), Dalton's Law of Partial Pressure, kinetic-molecular theory and characteristics of an ideal gas. While these lessons were originally designed to correlate to the performance indicators of the New York State Chemistry - Physical Setting curriculum, the components of this lesson may be easily used in other chemistry or physical science courses as well. The components this unit addresses in the NGSS Standards and Common Core Standards are indicated at the end of this description.
Many documents are included in both word and pdf to allow editing for specific teacher needs. Most of the activities in this package are well suited to distance learning environments.
The specific contents of the learning package includes the following items (the page count for these items are actual student handouts as answer key page counts are not included):
-- Marzano self assessment scale including learning goals and Common Core Reading/Writing and Mathematics Standards for students specific to this unit (3 pages)
-- Cloze notes handout for students with learning goals and Common Core Standards to accompany lesson PowerPoint (11 pages)
-- 58 slide PowerPoint to accompany the cloze notes
-- Specific Heat and Temperature Conversions Worksheet with answer key in PowerPoint format (4 pages/22 questions)
-- Phase Change worksheet with answer key (1 page/8 questions)
-- Heating Curve Worksheet with with answer key (1 page/8 questions)
-- Heating Curve quiz with answer key (1 page/4 questions)
-- Phase Changes and Gas Laws worksheet with answer key (4 pages/44 questions)
-- Partial Pressures and Gas Laws worksheet (answer key at the end of worksheet) (3 pages/18 questions)
-- Phase Change and Gas Law calculations quiz with answer key (2 pages/6 questions)
-- Review sheet for the Physical Behavior of Matter with answer key (3 pages/18 questions)
-- Physical Behavior of Matter Exam with answer key (8 pages/30 questions)
-- Links to other resources and lab activities pertaining to this unit (1 page)
Learning Goals (Local)
Upon the completion of this unit the student will be able to:
1. distinguish between heat and temperature in terms of molecular motion and amount of matter.
2. convert temperatures in Celsius and kelvin degrees.
3. calculate the heat (q=mc∆T or q=mHf or q=mHv ) involved in a phase or temperature change for a given sample of matter.
4. differentiate between the three types of heat calculations.
5. qualitatively interpret heating and cooling curves in terms of changes in kinetic energy and potential energy, heat of vaporization, heat of fusion and phase changes.
6. identify the melting point and boiling points from a heating/cooling curve.
7. explain phase change in terms of energy and intermolecular distance.
8. describe the states of the elements at STP.
9. identify what components in the gas law formula are required to solve a gas law problem.
10. solve for the amount of temperature, pressure, or volume using the gas law formula.
11. explain the gas laws in terms of kinetic molecular theory.
12. propose possible gases (ideal gases) which behave according to the kinetic theory.
13. calculate the number of liters, molecules or moles of gases given the following conditions using: 1 mole = 6.02 X 1023 molecules = gfm = 22.4 L
14. compare the entropy of different phases of matter.
15. summarize properties of gases, liquids and solids.
16. use a simple particle model to differentiate among properties among properties of solids, liquids, and gases.
NGSS Standards
Students who demonstrate understanding can:
HS-PS3-1. Create a computational model to calculate the change in the energy of one component in a system when the change in energy of the other component(s) and energy flows in and out of the system are known.
HS-PS3-2. Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-3. Design, build, and refine a device that works within given constraints to convert one form of energy into another form of energy.
HS-PS3-4. Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system (second law of thermodynamics).
STAAR Standards
Readiness Standards
C.9.A describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as described by Boyle’s law, Charles’ law, Avogadro’s law, Dalton’s law of partial pressure, and the ideal gas law.
Supporting Standards
C.4.C compare solids, liquids, and gases in terms of compressibility, structure, shape, and volume
C.9.B perform stoichiometric calculations, including determination of mass and volume relationships between reactants and products for reactions involving gases
C.9.C describe the postulates of kinetic molecular theory
C.11.A understand energy and its forms, including kinetic, potential, chemical, and thermal energies
C.11.B understand the law of conservation of energy and the processes of heat transfer
C.11.D perform calculations involving heat, mass, temperature change, and specific heat
C.11.E use calorimetry to calculate the heat of a chemical process
Common Core State Standards Connections:
ELA/Literacy
RST.11-12.1 Cite specific textual evidence to support analysis of science and technical texts, attending to important distinctions the author makes and to any gaps or inconsistencies in the account.
WHST.9-12.9 Draw evidence from informational texts to support analysis, reflection, and research.
Mathematics
MP.2 Reason abstractly and quantitatively.
MP.4 Model with mathematics.
HSN-Q.A.1 Use units as a way to understand problems and to guide the solution of multi-step problems; choose and interpret units consistently in formulas; choose and interpret the scale and the origin in graphs and data displays.
HSN-Q.A.2 Define appropriate quantities for the purpose of descriptive modeling.
HSN-Q.A.3 Choose a level of accuracy appropriate to limitations on measurement when reporting quantities.
Terms of Use
Purchase of the product is for classroom use by the purchaser only. It is a violation for individuals, schools, and districts to redistribute, sell, or post this item on the Internet or to other individuals.
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
View my complete High School Complete Chemistry Course